Products
Tono-Pen® & Ocu-Film® +
For hassel free & accurate hand held IOP readings
The award winning Reichert Tono-Pen AVIA tonometer is an easy to use, handheld instrument that provides IOP readings that correlate strongly with Goldmann tonometry. Tono-Pen AVIA has a lightweight, ergonomic design and advanced electronic measurement technology that enables operators to take fast and accurate IOP measurements with minimal training. Tono-Pen AVIA is powered by a long-life POWERCEL battery pack, good for over 2,500 measurements. Tono-Pen AVIA, like the Tono-Pen XL, uses sanitized Ocu-Film Tip Covers to minimize the risk of patient cross contamination. Tono-Pen AVIA is made in the USA.
Genuine Brand Ocu-Film+ tip covers are the only Reichert approved covers for use with Tono-Pen tonometers. Ocu-Film+ tip covers protect your Tono-Pen tonometers from dust and fluids, and help protect patients from the risks of cross contamination. Ocu-Film+ tip covers are 100% hand inspected by trained technicians and then sanitized. They are available in bulk bags or individually packaged in polyvinyl pouches.
+Learn more about Tono-Pen® & Ocu-Film® +iPac® Pachymeter
The Reichert iPac Pachymeter is the most advanced handheld pachymeter available. This easy-to-use, lightweight, hand held instrument includes all of the features that you have been searching for in other hand held pachymeters, but have been disappointed with your options. The iPac will impress you with features you have only dreamed about... Bluetooth wireless connectivity, one-button navigation, rotating color LCD screen and rechargeable lithium ion battery.+Learn more about iPac® Pachymeter
Reichert® 7CR Auto Tonometer + Corneal Response Technology®
The "Glaucoma Tonometer" with Corneal Response Technology® and IOPcc The Reichert 7CR from Reichert Technoligies utilizes a patented bi-directional applanation process to characterize the biomechanical properties of the cornea and reduce their impact on the IOP measurement. This measurement, referred to as Corneal Compensated IOP (IOPcc) is minimally affected by corneal visco-elastic properties, thickness, or surgical procedures such as LASIK or PRK. As such, IOPcc is a better indicator of glaucoma risk than other methods of tonometry, including Goldmann Applanation. +Learn more about Reichert® 7CRReichert® Ocular Resonse Analyzer®
The Ocular Response Analyzer (ORA) is the only instrument capable of measuring Corneal Hysteresis (CH); an indication of the biomechanical properties of the cornea. This information is different from thickness or topography, which are geometrical attributes of the cornea. Corneal Hysteresis represents a tissue property, which provides more comprehensive information about ocular biomechanics. There are over 400 publications about Ocular Response Analyzer in the peer-reviewed literature
The Corneal Hysteresis measurement has been shown to be consistently associated with, or predictive of, rate of glaucoma visual field progression. In addition, corneal biomechanics are the primary influence on tonometer accuracy. Ocular Response Analyzer's ability to measure Corneal Hysteresis enables the device to provide corneal compensated IOP, called IOPcc, which has been proven to be less influenced by corneal properties than Goldmann or other methods of tonometry.
+Learn more about Reichert® ORAReflex Ultrasound Biomicroscope
Ocular ultrasonography is an important adjunct for the clinical assessment of various ocular and orbital diseases. The Reichert Reflex Ultrasound Biomicroscope enables operators to easily image the anterior and posterior segments of the eye; providing important information not possible with clinical examination alone.
Users can now image pathologies, which may be obstructed by opacities or ocular structures, that OCT technology cannot. The easy to use touch screen interface provides effortless use of analysis tools, while the unique probe design allows the operator to examine the patient in sitting or reclined positions, without the need for a traditional water bath.
+Learn more about Reflex Ultrasound BiomicroscopeLasersoft
The Future in Refractive Laser Technology
210 nm WAVELENGTH:
PASSES THROUGH WATER AND BSS
Corneal Fluid and Humidity Changes have no effect on the ablation rate,No Nomograms, Gaussian Beam, Collimated Surgical Beam
SCANNING SPOT DELIVERY: SMOOTH ABLATION PROFILE
Small Spot Size (0.2 mm), High Repetition Rate (up to ~4 kHz), High-Speed Scanner
FAST VIDEO EYE TRACKING (1050 Hz) – FAST ANALOG EYE TRACKER (5 kHz)
Video Eye Tracker with a Latency of less than 1.0 ms – Analog Eye Tracker with a Latency of less than 0.2 ms
COAXIAL PATIENT FIXATION – OPHTHALMIC SURGICAL MICROSCOPE / VIDEO IMAGING
Accurate Patient Alignment
INTELLIGENT SOFTWARE: TRUE CUSTOMIZED CORNEAL ABLATION – PRESBYOPIA
Programmable Aspherical, Q-value adjusted, Wavefront and Topography guided ablations, Presbyopia.
ADVANCED TECHNOLOGY: SOLID STATE LASER PLATFORM
Reliable, Low Maintenance Cost, No Toxic Gas, Upgrade for Future Surgical Procedures
+Learn more about katanalaserMicrokeratome
MED-LOGICS, Inc. was founded on September 17, 1992 to focus on providing surgeons with the tools they need to optimize their patient outcomes. MED-LOGICS became the first company to receive 510(k) clearance from the FDA for marketing a replacement microkeratome blade on December 9, 1996.We have been appointed as distributor of Med-Logics, Inc. USA for WEST & EASTERN Part of INDIA and SAARC Countries for their Lasik, Dsaek & Cataract products.
LASIK: ML7 Microkeratome, Blades, Tubing & Heads.
DSAEK: ML7 Donor Cornea System, Nitro Donor Cornea System, Moria CBM Upgrades
Cataract: CATAPULSE a lens removal system (coming soon)
+Learn more about Med-Logics Inc., USA
MERILAS 532 α
The green 532 nm laser for
MERILAS 532α – the smart choice Small, portable, and light weight unit Ease of use due to graphical user interaction «Fast Start» software for shortest possible setup time Quiet operation (0 dB) without the noise of a cooling fan User interface detachable for user convenience with glass touch screen, easy to clean and highly resistan Low maintenance requirements and long service life due to diode pump technology Compatible with widest range of delivery option.
+Learn more about MERILAS 532αMICRORUPTOR V
YAG Laser
he MICRORUPTOR V is available in two versions:
MICRORUPTOR V footpedal
MICRORUPTOR V STAR
with joystick fire Dual rotating aiming beam for safe and precise operation.This beam can be adjusted easily for brightness and speed of rotation. It can even be turned off (for examination) or stopped if desired.MICRORUPTOR V – the Nd:YAG laser for Capsulotomy
Iridotomy Pupillary Membranectomy Ergonomic design for full patient- and doctor access Fully wheelchair accessible motorised table Left and right hand operation controls on column Easy to read menu on control panel Posterior and anterior defocusing of the position of the treatment focal plane at optimal 0.4mm Optimal setting for Iridotomy (repetition rate matches the «Eigenfrequency») Easy to use double and triple bursts for Iridotomy Integrated HAAG-STREIT BQ 900® slit lamp, the finest Swiss optics for optimal view and examination Compatible with most HAAG-STREIT BQ 900® accessories Unique rotating aiming beam for precise and safe focussing Easy to upgrade to a combination system with MERILAS 532α Green Laser Photocoagulator.
+Learn more about MICRORUPTOR VαSmartscope PRO - Feature Highlights
- Superior image quality fulfills ISO 10940 fundus camera standard
- Non-mydriatic eye fundus examination through undilated pupil
- Field of view 40 degrees
- 9 internal fixation targets for peripheral imaging
- Accurate and silent Autofocus and shoot
- WiFi for easy image transfer to any PC, laptop, tablet or mobile device
- Slit Lamp Adapter for any Slit Lamp with patient head rest
- Optomed Workstation software with DICOM for image viewing, sharing and archiving
- Low flash light intensity
- Smartscope ES2 module for high quality cornea imaging
STAAR PHAKIC IOLs (Visian ICL/TICL)
The Visian ICL (Implantable Collamer Lens), created by STAAR® Surgical Company, is a phakic intraocular lens (IOL) for the treatment of myopia and other refractive errors. The Visian TICL, a toric model of the lens, treats myopia with astigmatism. We have been appointed as stockiest for INDIA and also promoting the same as distributor in Western and Eastern part of INDIA.+Learn more about the STAAR ICL
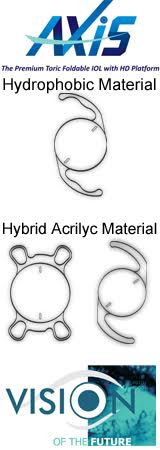
Revive Diffractive Multifocal Foldable Lens (Hybrid Acrylic Material)
Omni Lens Pvt. Ltd is a professionally managed & fastest growing IOL manufacturer company of INDIA. OMNI manufactures wide range of Foldable IOLs from US FDA approved raw material imported from USA through its state-of- the-art facility. We are promoting OMNI products within INDIA as sole distributor.+Learn more about OMNI
Crystalens® Intraocular Lens (IOL)
Bausch +Lomb Surgical provide surgeons with the tools and technology to perform all the latest techniques for the treatment of cataracts and vitreoretinal conditions.Crystalens AO – Accomodative IOL
Envista – Glistening FREE Hydrophobic Foldable IOL
Akreos MIL/AO/ADPAT – Acrylic Foldable IOL for MICS
Sofport AO/Soflex SE – Advance Optics (AO) Aspheric Lens
Stellaris Enhancement System - The Premium Platform for Advanced Lens Surgery. The Stellaris Vision Enhancement System is designed to complement your surgical technique and exceed your patient's expectations. It enables sub-2 mm lens surgery through a flexible, hybrid approach to fluidics and advanced, ultra-efficient cutting dynamics.
+Learn more about the B&L Surgical
BLUMINATOR ® Ophthalmic Illuminator
The Eidolon Bluminator's patented LED-based design revolutionizes examinations of the cornea. The Bluminator produces a bright, spectrally pure beam of blue light, which causes foreign bodies, abrasions, and other epithelial defects to fluoresce vividly during examinations with fluorescein. The blue beam produced is precisely the wavelength required to maximally excite the dye. Since the magnitude of the fluorescence is proportional to both the intensity and spectral purity of the light, the Bluminator yields a clearer, more detailed view of the eye than ever before. The view is enhanced by a 7X magnified image to facilitate detection and diagnosis of corneal injuries and irregularities, streamline contact lens fittings, and speed up treatment. It is an indispensable addition to the medical toolkit.Amazing fluorescent effect means you can see details with the Bluminator that you might miss with a conventional slit lamp
• Extremely lightweight, ruggedly built, and field tested
• Powered by non-rechargeable batteries that provide 3 - 5 year lifespan with normal use
• Suits every examination venue, including office, hospital, emergency departments, and missions
• White LED version offered for general examination
• New! BLUMINATOR Darkfield (Blue LED / Yellow Filter Lens) available

Nitro Donor Cornea System
The Nitro TM DSAEK System provides advanced performance specifications, which include the following:
- Console with the following requirements:
- Nitrogen powered Console. Does not need electricity in any way to perform a procedure
- Built-in Artificial Anterior Chamber pressure meter — the only way to verify and validate the pressure to the cornea in the AAC. Without this feature, the user cannot create accurate nomograms and help preserve endothelial cells by using lower pressure settings
- Eliminates unneeded components from system that were intended for LASIK systems — thus reducing the demands on maintenance and subsequent costs
- Linear movement head/vacuum ring system with the following requirements
- Linear bearing to minimize forward and reverse friction throughout the transition process
- Reusable 100, 350, 400, 450 and 500 micron DSAEK heads — accurate to within 5 microns of the projected depth of cut
- Heads manufactured from stainless steel 440 to minimize possibility of damage and wear
- Heads coated with 3 microns of surgical grade chromium to increase lubricity and smooth operation
- Eliminates meniscus cuts caused by rotating head action across the cornea
- No need to reposition the cornea for a second cut that can be required by systems that use the rotating head action
- Calibrated Blades (CLB®) available from -30 microns to +20 microns in 10 micron increments to allow compensation for thicker or thinner corneas, allowing consistent dimensional cutting, and manufactured to a tolerance of +/- 5 microns in the projected depth of cut
- One-stopcock tubing for single-operator convenience
MED-LOGICS is dedicated to improving patient outcomes through advanced designs.
LASIK: ML7 Microkeratome, Blades, Tubing & Heads.DSAEK: ML7 Donor Cornea System, Nitro Donor Cornea System, Moria CBM Upgrades
Cataract: CATAPULSE a lens removal system (coming soon)
+Learn more about Med-Logics Inc., USA
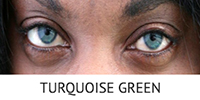
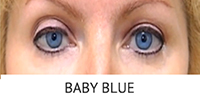
What is BrightOcular?
BrightOcular is a one-piece foldable ophthalmicgrade silicone anterior chamber intraocular implant that can be used in cases of iris abnormalities in patients such as ocular albinism, coloboma, total/partial aniridia as well as heterochromia and iris atrophy.
BrightOcular implants are able decrease photophobia and project the form of a healthy iris for patients with irregular iris appearance. The implant is offered in different colors to allow patients to select one that is best suited for them.
Who is BrightOcular for?
BrightOcular is for individuals diagnosed with ocular albinism, coloboma, aniridia, heterochromia, and iris atrophy and other iris-related abnormalities. In most of these conditions, light sensitivity is seen due to the deficiency of the iris’ natural light blockage role. Especially in albinos, BrightOcular can help to avoid premature cataract and retinal pathology development and may even play a role in reducing nystagmus, which is a major factor affecting visual acuity. Where a damaged, irregular iris may exist, BrightOcular can project a healthy iris that can play a role in relieving photosensitivity.
Why choose BrightOcular?
The benefits of BrightOcular include:
- hinner than existing phakic lenses
- Free flow of humour aqueous as a result of unobtructed and open visual axis
- Invented in the United states, the patented structure
- Ensures stability
- Minimizes pressure variation
- Maximizes flexibility
- Reduces contact surface between implant and iris by half
- Structural considerations translate to reduction and prevention of
- Post-operative inflammation
- Epithelial cell loss
- Glaucoma
- Simple, short, and painless procedure
How is implantation done?
Implantation is achieved using existing safe and effective FDA-certified intraocular lens implantation techniques. The techniques are equivalent to those of cataract procedures or ICL implantation. The surgery takes approximately 15 – 20 minutes per eye.
Implantation requires a preparation stage where topical anesthesia is applied. Next, the corneal limbus is incised followed finally by the injection of the implant into the anterior chamber.
Pre-Operative Evaluation/Contraindications
The following evaluations must be taken into consideration when determining suitability of procedure:
| Evaluation | Parameter |
|---|---|
| Intraocular Pressure | No greater than 22 mmHg (Must be evaluated with corneal thickness and optic disk examination to rule out normal tension glaucoma. Suspicious cases should have visual field examination and/or OCT) |
| Anterior Chamber Depth of Eye | > 2.50 |
| White-to-White Corneal Diameter | To determine implant size: • WTW ≥ 11.7, use 12.5 mm implant • WTW < 11.7, use 12.0 mm implant |
| Gonioscopy of Anterior Chamber Angle | Grade 4 Open |
| Peripheral Retinal Examination | No peripheral retinal changes/lesions present |
| Axial Length of Eye | < 25 mm for low risk of retinal detachment |
| Endothelium Cell Count, Specular Microscopy (>40 years old) | >2000/mm² |